5.2 Formation of ionic bonds
Ø Ionic Bond
Ø Formation of cation
Ø Ionic Bond
- Ionic bonds are formed by one atom transferring electrons to another atom to form ions.
- Metal atoms – donate electron formed cation
- Non-metal atoms – accept electron formed anion
- Ions of opposite charge will attract one another by strong electrostatic force, thus creating an ionic bond.
- Ionic bond is also known as electrovalence bond.
Ø Formation of cation
- When an atom donates electrons, it becomes positively charged as there are more protons than electrons in it.
- A positively charged ion or cation is obtained.
- M → Mn+ + ne
- Example:
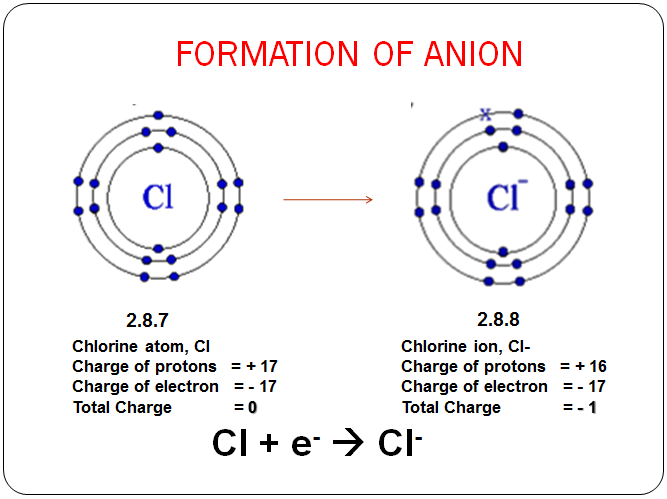
Ø Formation of anion
- When an atom accepts electrons, it becomes negatively charged as there are more electrons than protons in it.
- A negatively charged ion or anion is obtained
- A + ne → An-
- Example:
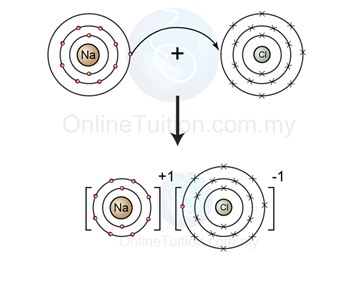
Ø Example 1: Ionic Bonding between Group 1 Metal and Group 17 Element
- Figure above shows the illustration of the formation of ionic bond between a sodium atom (group 1 metal) and a chlorine atom (group 17 element).
- The electron arrangement of sodium atom is 2.8.1, which is not octet and hence not stable.
- To achieve octet electrons arrangement, the sodium atom donate/release one electron and form sodium ion.
- The electron arrangement of chlorine atom is 2.8.7, which is also not octet and hence not stable, too.
- To achieve octet electrons arrangement, the chlorine atom receives one electron from sodium atom and form a chloride ion.
- The sodium ion and the chloride ion carry opposite charge, hence they attract each other and form an ionic bond between each other.
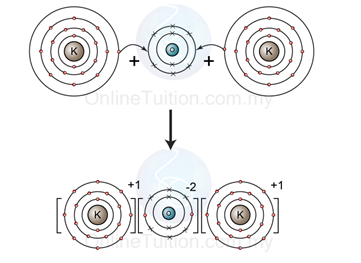
Ø Example 2: Ionic Bonding between Group 2 Metal and Group 16 Element
- Figure above shows the illustration of the formation of ionic bond between two potassium atoms (group 1 metal) and an oxygen atom (group 16 element).
- The electron arrangement of potassium atom is 2.8.8.1, which is not octet and hence not stable.
- To achieve octet electrons arrangement, the potassium atom donate/release one electron and form potassium ion.
- The electron arrangement of oxygen atom is 2.6, which is also not octet and hence not stable, too.
- To achieve octet electrons arrangement, the oxygen atom receives two electrons from potassium atoms and form an oxide ion.
- The potassium ions and the oxide ion carry opposite charge, hence they attract each other and form two ionic bond between the ions.
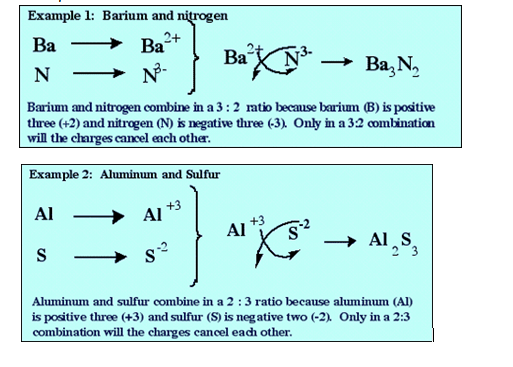
Ø Determining the ionic compound formulae
- Formulae of an ionic compound.
- Metal atoms will release their valence electrons to achieve the stable electron arrangement as in the inert gases.
- Non-metal atoms will accept electrons in order to achieve the stable electron arrangement of the inert gases.
- For cations Mb+ and anions Xa-, the formula of an ionic compound formed between them is written as MaNb.
- The total positive charge of the cation must be equal to the negative charge of the anion in an ionic compound.
- Hence, the formula of an ionic compound formed between them can also be derived as follows :
- aMb+ + bXa- → MaNb
- Examples: